Sections in This Article
Note Depending on your Admin's configuration, object, field, and section labels, lifecycle states, and workflows may differ from the general information on this page. Refer to your organization's business processes for guidance.
About Masked Safety Reports
When you configure a Submission or Distribution, you can choose to protect confidential or blinded data in the generated ICSR, such as all of a patient’s identifiable and health information, only identifiable information, or unblinded study data.
By setting content confidentiality preferences for Partner Distributions, you can generate masked distributions for partner organizations while continuing to generate unblinded ICSRs for agency submissions.
There are masking settings specific to Partner Distribution families. Each setting masks or hides certain information on generated regulatory reports or E2B Transmissions.
The following fields control safety report masking:
- Patient Content Protection (
patient_content_protection__v) - Exceptions to Patient Content Protection (
exceptions_to_patient_content_protection__v) - Study Content Protection (
study_content_protection__v)
See the following table for an overview of how to generate masked safety reports for Transmissions:
| Transmission Type | Set Up |
|---|---|
| Distribution |
|
| Submission |
|
In addition to the masking field settings for Submissions and Distributions, your Admin can optionally enable the system to mask dosage to study products and mask the reporter for postmarket non-literature Cases.
Your Admin may also configure unmasked export of open-label Products within blinded Studies on blinded CIOMS I and FDA 3500A forms.
Prerequisite
Ensure that your Vault has the Masked Content Distributions feature enabled.
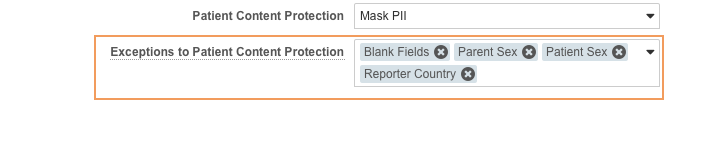
Patient Content Protection
Use the Patient Content Protection field to configure masking for personally identifiable information (PII).
You can configure masking exceptions for Patient Content Protection (PCP) using the Exceptions to Patient Content Protection field.
Mask Personal Identifiable Information (PII)
Vault Safety masks the following fields when you select Mask PII while configuring a Partner Distribution Rule:
Note When the Date of Birth field is masked, the Age and Age Group information will be transmitted during E2B transmission.
| Object | Field | FDA 3500A/CIOMS I Mask Indicator | E2B(R3) Mask Indicator | E2B(R2) Mask Indicator |
|---|---|---|---|---|
| Case Contact | Title (title_value__v) |
***** | MSK | PRIVACY |
| Case Contact | First Name (firstname_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Middle Name (middlename_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Last Name (lastname_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Organization (organization_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Department (department_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Street (street_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Postal Code (postalcode_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Telephone (telephone_value__v) |
***** | MSK | Left Blank1 |
| Case Contact | Fax (fax__v) |
***** | MSK | Left Blank1 |
| Case Contact | Email Address (email_address__v) |
***** | MSK | Left Blank1 |
| Case Version | Reporter First Name (reporter_first_name__v) |
***** | MSK | PRIVACY |
| Case Version | Reporter Last Name (reporter_last_name__v) |
***** | MSK | PRIVACY |
| Case Version | Patient ID (patient_id_value__v) |
***** | MSK | PRIVACY |
| Case Version | Gender (gender_value__v) |
Left blank | MSK2 | Left Blank1 |
| Case Version | MRN - GP (mrn_gp_value__v) |
***** | MSK | Left Blank1 |
| Case Version | MRN - Specialist (mrn_specialist_value__v) |
***** | MSK | Left Blank1 |
| Case Version | MRN - Hospital (mrn_hospital_value__v) |
***** | MSK | Left Blank1 |
| Case Version | MRN - Investigation (mrn_investigation_value__v) |
***** | MSK | Left Blank1 |
| Case Version | Date of Birth (dob_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Date of Birth (normalized) (dob_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Last Menstrual (last_menstrual_idate__v) |
**-***-**** | MSK2 | Left Blank1 |
| Case Version | Last Menstrual (normalized) (last_menstrual_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Medical History Text (medical_history_text__v) |
***** | MSK2 | Left Blank1 |
| Case Version | Date of Death (dod_idate__v) |
**-***-**** | MSK2 | Left Blank1 |
| Case Version | Date of Death (normalized) (dod_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Product | Date Implanted (date_implanted__v) |
**-***-**** | MSK | Left Blank1 |
| Case Product | Date Explanted (date_explanted__v) |
**-***-**** | MSK | Left Blank1 |
| 1. This is either not a valid element or nullFlavor for E2B(R2). If this is a valid element, the element will not be transmitted in place of being masked. | ||||
| 2. When exporting an EMA E2B(R3) with a transmission destination of EMA, this field is not masked. If the field contains a manually entered MSK value, then the field is omitted from the transmission. | ||||
Exceptions to Patient Content Protection
When using PCP, you can select PII fields to leave unmasked.
You can configure masking exceptions for the following fields using the Exceptions to Patient Content Protection dropdown menu:
| Exception | Unmasked Field | Object |
|---|---|---|
| Blank Fields | - | - |
| Parent Sex | Gender (gender_value__v)1 |
Case Version (Parent) |
| Patient Sex | Gender (gender_value__v)1 |
Case Version |
| Reporter Country | Country (country_value__v) |
Case Contact (Reporter) |
| 1. When exporting an EMA E2B(R3) with a transmission destination of EMA, this field is not masked. If the field contains a manually entered MSK value, then the field is omitted from the transmission. | ||
If you do not see the Exceptions to Patient Content Protection field, your Admin may need to Enable Configurable Field Masking.

Select any number of fields from the dropdown menu to leave unmasked.
E2B Mask (Includes Personal Identifiable Information)
Vault Safety masks all of the PII fields listed above, as well as the fields in the following table when you select E2B Mask while configuring a Partner Distribution Rule.
Note For FDA VAERS E2B(R3) exports, the following fields are not masked, as MSK is not a valid nullFlavor setting for these E2B data elements:
- FDA.D.13 - Pregnant at Vaccination
- FDA.D.1f - Street Line 2 (Patient)
- FDA.C.2.r.2.3 - Street Line 2 (Reporter)
| Object | Field | FDA 3500A/CIOMS I Mask Indicator | E2B(R3) Mask Indicator | E2B(R2) Mask Indicator |
|---|---|---|---|---|
| Case Contact | Title (title_value__v) |
***** | MSK | PRIVACY |
| Case Contact | First Name (firstname_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Middle Name (middlename_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Last Name (lastname_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Organization (organization_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Department (department_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Street (street_value__v) |
***** | MSK | PRIVACY |
| Case Contact | Street - Line 2(street_line_2_value__v) |
***** | MSK | Left Blank1 |
| Case Contact | Postal Code (postalcode_value__v) |
***** | MSK | PRIVACY |
| Case Contact | County (county_value__v) |
***** | MSK | Left Blank1 |
| Case Contact | Country (country_value__v) |
***** Form checkbox is blank (G.3) |
MSK | Left Blank1 |
| Case Contact | Telephone (telephone_value__v) |
***** | MSK | Left Blank1 |
| Case Contact | Fax (fax__v) |
***** | MSK | Left Blank1 |
| Case Contact | Email Address (email_address__v) |
***** | MSK | Left Blank1 |
| Case Contact | City (city_value__v) |
***** | MSK | PRIVACY |
| Case Contact | State / Province (state_province_value__v) |
***** | MSK | PRIVACY |
| Case Version | Reporter First Name (reporter_first_name__v) |
***** | MSK | PRIVACY |
| Case Version | Reporter Last Name (reporter_last_name__v) |
***** | MSK | PRIVACY |
| Case Version | Patient ID (patient_id_value__v) |
***** | MSK | PRIVACY |
| Case Version | Gender (gender_value__v) |
Left blank | MSK | Left Blank1 |
| Case Version | MRN - GP (mrn_gp_value__v) |
***** | MSK | Left Blank1 |
| Case Version | MRN - Specialist (mrn_specialist_value__v) |
***** | MSK | Left Blank1 |
| Case Version | MRN - Hospital (mrn_hospital_value__v) |
***** | MSK | Left Blank1 |
| Case Version | MRN - Investigation (mrn_investigation_value__v) |
***** | MSK | Left Blank1 |
| Case Version | Date of Birth (dob_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Date of Birth (normalized) (dob_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Last Menstrual (last_menstrual_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Last Menstrual (normalized) (last_menstrual_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Medical History Text (medical_history_text__v) |
***** | MSK | Left Blank1 |
| Case Version | Date of Death (dod_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Date of Death (normalized) (dod_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Event Onset (event_onset_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Event Onset (normalized) (event_onset_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Race (race__v) |
***** | MSK | Left Blank1 |
| Case Version | Ethnicity (ethnicity__v) |
***** | MSK | Left Blank1 |
| Case Version | Pregnant at Vaccination (pregnant_at_vaccination__v) |
***** | MSK | Left Blank1 |
| Case Product | Date Implanted (date_implanted__v) |
**-***-**** | MSK | Left Blank1 |
| Case Product | Date Explanted (date_explanted__v) |
**-***-**** | MSK | Left Blank1 |
| Case Version | Product Generic Name (generic_name__v) |
***** | MSK | Left Blank1 |
| Case Version | MPID Version (mpid_version__v) |
***** | MSK | Left Blank1 |
| Case Adverse Event | Onset (onset_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Adverse Event | Onset (normalized) (onset_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Adverse Event | Cessation (resolved_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Adverse Event | Cessation (normalized) (resolved_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Adverse Event | Duration Duration (unit) ( duration_number__v)) |
***** | MSK | Left Blank1 |
| Case Drug History | Start Date (startdate_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Drug History | Start Date Normalized (startdate_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Drug History | End Date (enddate_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Drug History | End Date Normalized (end_date_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Medical History | Start Date (startdate_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Drug History | Start Date Normalized (startdate_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Drug History | Continuing (continuing_value__v) |
***** | MSK | Left Blank1 |
| Case Drug History | End Date (enddate_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Drug History | End Date Normalized (enddate_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Product Dosage | First Administration (firstadmin_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Product Dosage | First Administration (normalized) (firstadmin_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Product Dosage | Last Administration (lastadmin_idate__v) |
**-***-**** | MSK | Left Blank1 |
| Case Product Dosage | Last Administration (normalized) (lastadmin_normalized__v) |
**-***-**** | MSK | Left Blank1 |
| Case Product Dosage | Duration (number) Duration (unit) ( duration_number__v)) |
**-***-**** | MSK | Left Blank1 |
| 1. This is either not a valid element or nullFlavor for E2B(R2). If this is a valid element, the element will not be transmitted in place of being masked. | ||||
Study Content Protection
Use the Study Content Protection field to configure masking for unblinded information on Study Cases.
Mask Unblinded Content
Vault Safety masks the following fields when you select Mask Unblinded Content while configuring a Partner Distribution Rule:
Note When you select Mask Unblinded Content on a Spontaneous Case with Study Content Protection, the Case’s Product information will remain unblinded on CIOMS I and 3500A forms.
Tip By default, Product Dosage information is unmasked. However, your Admin can enable Mask Dosage in Blinded Distributions to apply with the Mask Unblinded Content option.
| Object | Field | FDA 3500A/CIOMS I Mask Indicator | E2B(R2) and (R3) Mask Indicator |
|---|---|---|---|
| Case Product Dosage | Batch/Lot Number (batchlot_number__v) |
Left Blank | Left Blank |
| Case Product | Product (Coded) (product_name_v) |
Blinded name or ***** if no blinded name | Blinded name or MSK if no blinded name |
| Case Product | Product (Reported) (product_reported__v) |
Blinded name or ***** if no blinded name | Blinded name or MSK if no blinded name |
| Case Product | MPID (mpid__v) |
***** | Left Blank |
| Case Product | MPID Version (mpid_version__v) |
***** | Left Blank |
| Case Product | PhPID (phpid__v) |
***** | Left Blank |
| Case Product | PhPID Version (phpid_version__v) |
***** | Left Blank |
| Case Product | Registration Number (registration_number__v) |
Left Blank | Left Blank |
| Case Product | Registration Country (registration_country__v) |
***** | Left Blank |
| Case Product | Registration Holder/Applicant (registration_mah__v) |
***** | Left Blank |
| Case Product | Organization (Organization__v) |
***** | Left Blank |
| Case Product | Expiration Date (expiration_date__v) |
***** | Left Blank |
| Case Product | Name (name__v) |
***** | Left Blank |
| Product | Manufacturer (manufacturer__v) |
***** | Left Blank |
| Case Product | Model Number (model_number__v) |
***** | Left Blank |
| Case Product | Catalog Number (catalog_number__v) |
***** | Left Blank |
| Case Product | Serial Number (serial_number__v) |
***** | Left Blank |
| Case Product | Lot Number (lot_number__v) |
***** | Left Blank |
| Case Product | Unique Identifier (unique_Identifier__v) |
***** | Left Blank |
| Case Product | Reprocessed/Reused (reprocessed__v) |
***** | Left Blank |
| Case Product | Reprocessor (reprocessor__v) |
***** | Left Blank |
| Case Product | Additional Information (additional_information_coded__v) |
***** | Left Blank |
| Case Product | Other Additional Information (additional_information_text__v) |
***** | Left Blank |
| Case Product | Blinded (blinded__v) |
***** | Left Blank |
| Product | Generic Name (generic_name__v) |
***** | Left Blank |
| Case Product Substance | Name (Reported) (name_reported__v) |
***** | Left Blank |
| Case Product Substance | TermID Version (termid_version__v) |
***** | Left Blank |
| Case Product Substance | TermID (termid__v) |
***** | Left Blank |
| Case Product Substance | Strength (Number) (strength_number__v) |
***** | Left Blank |
| Case Product Substance | Strength (Unit) (strength_unit__v) |
***** | Left Blank |
Dosage Masking for Study Products
By default, Product Dosage information is unmasked in generated reports for masked Distributions.
However, Vault Safety supports the option to mask Study Product dosage on E2B, CIOMS I, and FDA 3500A reports.
Enable Dosage Masking for Study Products
Your Admin can enable Mask Dosage in Blinded Distributions in your Vault.
If enabled, Study Product dosage masking applies to reports generated in the following conditions:
- Masked Distributions where Study Content Protection is set to "Mask Unblinded Content"
- Blinded report previews (manually generated from a blinded Study Case)
Masked Dosage Fields
If dosage masking is enabled, the following dosage information is masked:
- Dose
- Patient Route of Administration
- Dose Frequency
Note Dose Frequency is not displayed if the Frequency field is blank when generating a report.
Dosage Masking by Format
The following table outlines how dosage masking impacts different report types:
| Report Type | Dosage Masking |
|---|---|
| CIOMS I | Box 15 and 16 are populated with ***** |
| FDA 3500A | Box C.3 is populated with ***** |
| E2B(R3) | Dosage section G.k.4.r is omitted |
| E2B(R2) |
Dosage sections are omitted:
|
Reporter Masking for Postmarket Non-Literature Cases
Vault Safety provides a system-level setting to support masking reporter information in all outbound Submissions and Distribution for postmarket non-literature Cases. This setting is controlled from the Submission and Distribution Settings page in the Admin area.
The following conditions must be met to mask reporter fields for a Case:
- The Mask Reporter for PM Non-Literature Case system setting must be enabled in your Vault.
- The Case Report Type must correspond to a postmarket, non-literature report type:
- Postmarket: The Case must meet one of the following conditions to be considered "postmarket":
- Non-Study Report Type: The Case Report Type specifies a non-study report type. That is, the Report Type corresponds to a Controlled Vocabulary of "Spontaneous" (E2B Code of
1), "Other" (E2B Code of3), or "Not Available" (E2B Code of4). - Postmarket Study Case: The Case Report Type corresponds to a Study Controlled Vocabulary (E2B Code of
2) and the Case Study Type corresponds to a Controlled Vocabulary of "Individual Patient Use" (E2B Code of2) or "Other Study" (E2B Code of3).
- Non-Study Report Type: The Case Report Type specifies a non-study report type. That is, the Report Type corresponds to a Controlled Vocabulary of "Spontaneous" (E2B Code of
- Non-Literature: The Case Report Type field specifies a non-literature report type. That is, the option selected corresponds to a Report Type Controlled Vocabulary with the Literature field set to "No" or unspecified.
- Postmarket: The Case must meet one of the following conditions to be considered "postmarket":
Enable Reporter Masking System Setting
- Go to Admin > Settings > Submission and Distribution Settings.
Your Permission Set must have permission to View the Submission and Distribution Settings page to access this page. - Enable the Mask Reporter for PM Non-Literature Case field.
Note Once enabled, reporter masking applies to all postmarket non-literature Cases, regardless of the masking settings on the Transmission.
Masked Reporter Fields
The system masks the following information for Reporter-type Case Contacts when generating any report format:
Note If the Patient Content Protection field specifies 'E2B Mask' on a Transmission, the system masks the Reporter Country in addition to the fields below in the generated report.
| Object | Field |
|---|---|
| Reporter-type Case Contact | Title (title_value__v) |
| Reporter-type Case Contact | First Name (firstname_value__v) |
| Reporter-type Case Contact | Middle Name (middlename_value__v) |
| Reporter-type Case Contact | Last Name (lastname_value__v) |
| Reporter-type Case Contact | Organization (organization_value__v) |
| Reporter-type Case Contact | Department (department_value__v) |
| Reporter-type Case Contact | Street (street_value__v) |
| Reporter-type Case Contact | Postal Code (postalcode_value__v) |
| Reporter-type Case Contact | Telephone (telephone_value__v) |
| Reporter-type Case Contact | Fax (fax__v) |
| Reporter-type Case Contact | Email Address (email_address__v) |
Reporter Masking by Format
The following table outlines how reporter masking impacts different report types:
| Report Type | Reporter Masking |
|---|---|
| CIOMS I | Only the Reporter Qualification and Reporter Country fields are exported, if populated. If these fields are blank, no reporter information is transmitted. |
| FDA 3500A | Only the Reporter Qualification and Reporter Country fields are exported, if populated. If these fields are blank, no reporter information is transmitted. |
| E2B(R3) | Specified reporter fields are masked with MSK. Empty fields are not transmitted. |
| E2B(R2) | Specified reporter fields are masked with PRIVACY. Empty fields are not transmitted. |
