With the 23R2 release, SafetyDocs supports signal management, offering the ability to track signals as Product-Event Combinations, set PEC Periods, and record details of Safety Investigations.
Overview
To configure this feature in your Vault:
- Configure object layouts
- Configure the Reporting Period field on the Safety Investigation object
- Configure object workflows
- Configure object lifecycles
- Configure security
Configure Layouts
Configure the following object layouts:
- Product-Event Combination
- PEC Period
- Signal Source
- Safety Investigation
- Safety Investigation Reporting Period
Configure the Product-Event Combination Object Layout
To configure the Product-Event Combination object layout:
- Navigate to Admin > Configuration > Objects > Product-Event Combination > Layouts > Product-Event Combination Detail Page Layout.
- Drag the Primary MedDRA Term field from the Details section into the System section.
- To the Details section, add the
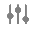 Primary MedDRA Term control field with the slider icon.
Primary MedDRA Term control field with the slider icon. - Configure the rest of the object layout as follows:
- Ensure the Details section contains the following fields in the following order:
- Primary Product Family
 Primary MedDRA Term
Primary MedDRA Term- Most Recent Disposition
- Most Recent Disposition Date
- Ensure the Product-Event Dispositions section contains the following default columns in the following order:
- Name
- Detection Date
- Reporting Period
- Primary Source
- Final Disposition
- Final Disposition Date
- Investigation Type
- Safety Investigation
- Ensure the System section contains the following fields in the following order:
- Combination Name
- Created By
- Created Date
- Primary MedDRA Term
- Last Modified By
- Last Modified Date
- Ensure the Documents section contains the following columns in the following order:
- Name
- Document Number
- Created By
- Created Date
- Ensure there is a Workflow Timeline section
- Ensure the Details section contains the following fields in the following order:
- Select Save.
Configure the PEC Period Object Layout
To configure the PEC Period object layout:
- Navigate to Admin > Configuration > Objects > PEC Period > Layouts > PEC Period Detail Page Layout.
- To the Details section, add the
 Primary MedDRA Term control field with the slider icon.
Primary MedDRA Term control field with the slider icon. - Configure the rest of the object layout as follows:
- Ensure the Details section contains the following fields in the following order:
- Product-Event Combination
- Primary Product Family
 Primary MedDRA Term
Primary MedDRA Term- Primary Source
- Reporting Period
- Investigation Type
- Detection Date
- Final Disposition
- Final Disposition Date
- Safety Investigation
- Ensure the Documents section contains the following columns in the following order:
- Name
- Document Number
- Type
- Subtype
- Classification
- Status
- Ensure there is a Workflow Timeline section
- Ensure the System section contains the following fields in the following order:
- Created By
- Created Date
- Last Modified By
- Last Modified Date
- Ensure the Details section contains the following fields in the following order:
- Select Save.
Configure the Signal Source Object Layout
To configure the Signal Source object layout:
- Navigate to Admin > Configuration > Objects > Signal Source > Layouts > Signal Source Detail Page Layout.
- Configure the object layout as follows:
- Ensure the Details section contains the Name field
- Ensure the Product-Event Dispositions section contains the following default columns in the following order:
- Name
- Detection Date
- Final Disposition
- Final Disposition Date
- Reporting Period
- Safety Investigation
- Investigation Type
- Primary Product Family
- Ensure there is a Workflow Timeline section
- Ensure the System section contains the following fields in the following order:
- Created By
- Created Date
- Last Modified By
- Last Modified Date
- Select Save.
Configure the Safety Investigation Object Layout
To configure the Safety Investigation object layout:
- Navigate to Admin > Configuration > Objects > Safety Investigation > Layouts > Safety Investigation Detail Page Layout.
- Configure the object layout as follows:
- Ensure the Details section contains the following fields in the following order:
- Product Family
- Primary Source
- Start Date
- Detection Date
- Priority
- Validation Outcome
- Validation Outcome Date
- Final Disposition
- Final Disposition Date
- Ensure there is a Workflow Timeline section
- Ensure the Product-Event Dispositions section contains the following default columns in the following order:
- Name
- Detection Date
- Final Disposition
- Final Disposition Date
- Reporting Period
- Safety Investigation
- Investigation Type
- Primary Product Family
- Ensure the Summary of Key Data section includes the Summary of Key Data field
- Ensure the Documents section contains the following columns in the following order:
- Name
- Document Number
- Type
- Subtype
- Classification
- Status
- Ensure the System section contains the following fields in the following order:
- Created By
- Created Date
- Last Modified By
- Last Modified Date
- Ensure the Details section contains the following fields in the following order:
- Select Save.
Configure the Safety Investigation Reporting Period Object Layout
To configure the Safety Investigation Reporting Period object layout:
- Navigate to Admin > Configuration > Objects > Signal Reporting Period > Layouts > Safety Investigation Reporting Period Detail Page Layout.
- Configure the object layout as follows:
- Ensure the Details section contains the following fields in the following order:
- Name
- Product Family
- Start Date
- End Date
- Ensure the Product-Event Dispositions section contains the following default columns in the following order:
- Name
- Detection Date
- Final Disposition
- Final Disposition Date
- Reporting Period
- Safety Investigation
- Investigation Type
- Primary Product Family
- Ensure the Safety Investigations section contains the following default columns in the following order:
- Name
- Detection Date
- Final Disposition
- Final Disposition Date
- Product Family
- Reporting Period
- Validation Outcome
- Ensure there is a Workflow Timeline section
- Ensure the System section contains the following fields in the following order:
- Created By
- Created Date
- Last Modified By
- Last Modified Date
- Ensure the Details section contains the following fields in the following order:
- Select Save.
Configure the Reporting Period Field
To configure the Reporting Period field on the Safety Investigation object:
- Navigate to Admin > Configuration > Objects > Safety Investigation > Fields.
- Select Reporting Period.
- Select Edit.
- Clear the Display in default lists and hovercards checkbox.
- Select Save.
Configure Object Workflows
Configure signal management object workflows. Use these steps as a starting point, but configure your workflows according to your business processes:
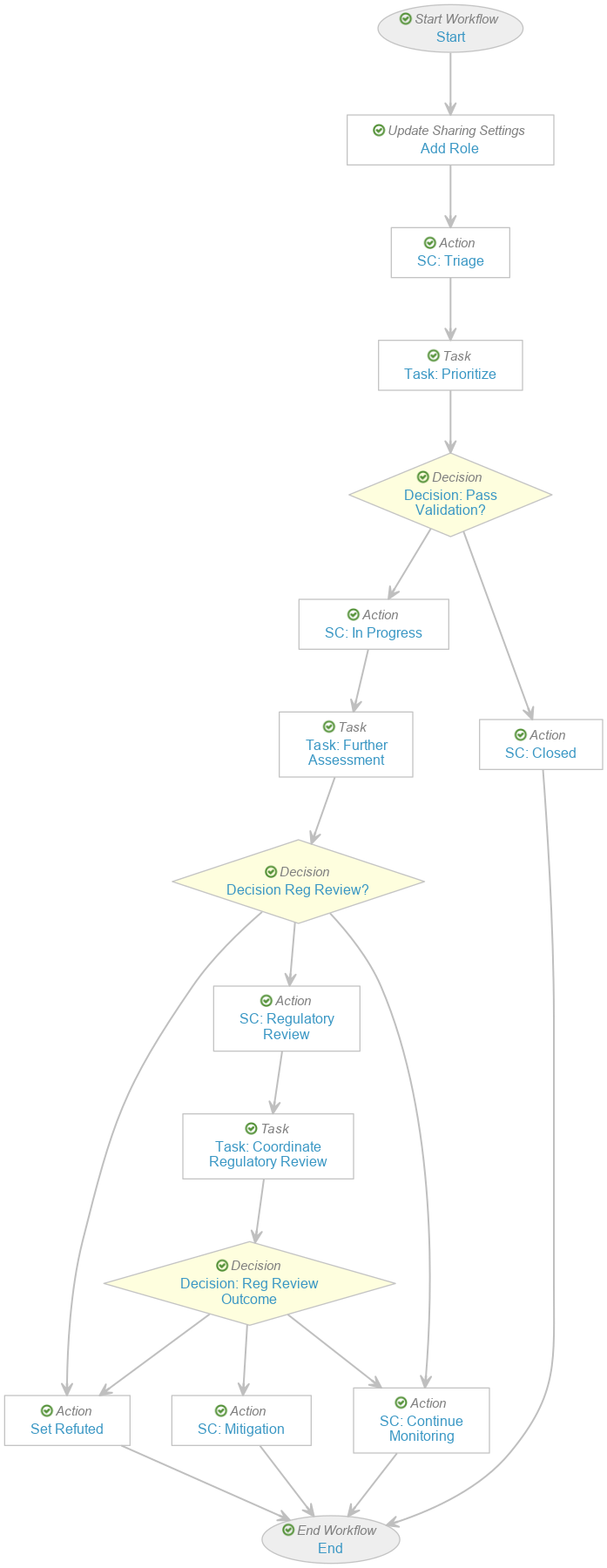
Create the Safety Investigation Workflow
To create the Safety Investigation Workflow:
- Navigate to Admin > Configuration > Workflows.
- Select Create.
- In the Create Workflow window, select Object Workflow.
- Select Continue.
- On the Create Workflow page, populate the fields as follows:
- Label: Enter
Safety Investigation Workflow - Lifecycle: Select Safety Investigation Lifecycle
- Label: Enter
- Select Save.
- In the Workflow Steps section, configure the workflow to include the following steps:
- Start start step
- Add Role update sharing settings step
- SC: Triage state change action step
- Task: Prioritize task step
- Decision: Pass Validation? decision step
- SC: In Progress state change action step
- Task: Further Assessment task step
- Decision: Reg Review? decision step
- Set Refuted action step
- SC: Regulatory Review state change action step
- Task: Coordinate Regulatory Review task step
- Decision: Reg Review Outcome decision step
- SC: Mitigation state change action step
- SC: Continue Monitoring state change action step
- SC: Closed state change action step
- End end step
- In the Status field, select Make configuration active.
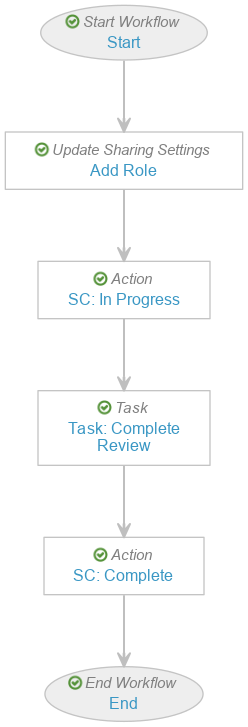
Create the Evaluate Disposition Workflow
To create the Evaluate Disposition Workflow:
- Navigate to Admin > Configuration > Workflows.
- Select Create.
- In the Create Workflow window, select Object Workflow.
- Select Continue.
- On the Create Workflow page, populate the fields as follows:
- Label: Enter
Evaluate Disposition Workflow - Lifecycle: Select Product-Event Disposition Lifecycle
- Label: Enter
- Select Save.
- In the Workflow Steps section, configure the workflow to include the following steps:
- Start start step
- Add Role update sharing settings step
- SC: In Progress state change action step
- Task: Complete Review task step
- SC: Complete state change action step
- End end step
- In the Status field, select Make configuration active.
Configure Object Lifecycles
To configure signal management object lifecycles:
- Configure actions on the Safety Investigation Lifecycle
- Configure actions on the Product-Event Disposition Lifecycle
- Configure a Safety Investigation Lifecycle Stage Group
Configure Actions on the Safety Investigation Lifecycle
To update the Safety Investigation Lifecycle object lifecycle to support the Safety Investigation Workflow, create the following:
| Lifecycle State | Type | Configuration |
|---|---|---|
| Open | User action |
|
| Monitoring | User action |
|
| Closed | Entry criteria |
|
Configure Actions on the Product-Event Disposition Lifecycle
To update the Product-Event Disposition Lifecycle object lifecycle to support the Evaluate Disposition Workflow, create the following:
| Lifecycle State | Type | Configuration |
|---|---|---|
| Active | User action |
|
| Complete | Entry criteria |
|
| Entry action |
|
Configure the Safety Investigation Lifecycle Stage Group
Create an object lifecycle stage group with the following settings:
- Stage Group Label: Enter
Safety Investigation Stages - Object Lifecycle: Select Safety Investigation Lifecycle
- Object Type: Select Base Safety Investigation
Then, create stages and define their states as follows:
| Stage | States |
|---|---|
| Signal Prioritization | Open |
| Signal Validation | Triage, Regulatory Review |
| Further Assessment | In Progress |
| Mitigation | Monitoring, Risk Mitigation |
| Complete | Complete |
Configure Security
Configure security settings to grant access to signal management features. Use the following settings as a starting point, but configure access according to your business processes:
- Create a permission set with the label
Signal Management - Create a security profile with the label
Signal Managementand assign to it the Signal Management permission set - Create a group with the label
Signal Managersand add the Signal Management security profile as an included security profile - Configure permissions for the Signal Management document type
- Grant permissions on the Signal Management permission set
Configure Document Type Permissions
To configure permissions for the Signal Management (signal_management__v) document type:
- Navigate to Admin > Configuration > Document Types.
- Select Signal Management (signal_management__v) and then in the All Actions menu, select Edit Details.
- Navigate to the Security tab.
- In the Create Document Permission field, add the following:
- Administrators
- Signal Managers
- Under Default Settings for New Documents, in the Editors field, add Signal Managers.
- Select Save.
Grant Permissions on the Signal Management Permission Set
To grant permissions to the Signal Management permission set:
- Grant Read object permissions on the following objects:
- Aggregate Reporting Group
- Case
- Case
- Parent Information
- Imported Case
- Child Information
- Case Access Group
- Case Adverse Event
- Case Assessment
- Case Assessment Expectedness
- Case Assessment Result
- All Case Cause of Death objects
- All Case Contact objects
- Case Diagnosis
- Case Drug History
- Case Drug History Substance
- Case Identifier
- Case Medical History
- Case Number
- All Case Product objects
- Case Product Device Code
- Case Product Dosage
- Case Product Indication
- All Case Product Registration objects
- Case Product Substance
- Case Relationship
- Case Signature
- Case Study Registration
- Case Test Result
- All Controlled Vocabulary objects
- Country
- Country State/Province
- Datasheet
- All Device Code objects
- Dictionary
- MedDRA Dictionary
- WHODrug Dictionary
- EDQM Standard Term
- IMDRF Device Code
- Document Lifecycle Stage Label
- Document Lifecycle State-Stage
- Document Type Detail
- Document Type Group
- Document Type Group Detail
- Document Usage
- Dose Form
- EDQM
- Emailed Document
- IMDRF Device Code
- Inactive Ingredient
- Inactive Ingredient Registration
- Inbox Item
- Language
- Language (ISO)
- Language Locale
- Literature Article
- Literature Database
- Literature Review
- Literature Search Term
- Local Reporting Details
- Local Reporting Details/Product Join
- Locale
- Localization
- Localized Case
- Localized Case Adverse Event
- Localized Case Assessment
- Localized Case Assessment Result
- Localized Case Cause of Death
- Localized Case Comment
- Localized Case Contact
- Localized Case Document
- Localized Case Drug History
- Localized Case Identifier
- Localized Case Medical History
- Localized Case Product
- Localized Case Product Device Code
- Localized Case Product Dosage
- Localized Case Product Indication
- Localized Case Product Substance
- Localized Case Test Result
- Localized Case Translation
- Localized Controlled Vocabulary
- Localized Country
- Localized Country State/Province
- Localized Dose Form
- Localized Organization
- Localized Product
- Localized Reason Omitted
- Localized Route of Administration
- Localized Study
- Localized Study Arm
- Localized Substance
- Localized Unit of Measurement
- Market Segment
- MedDRA
- MedDRA (Localized)
- MedDRA Criteria
- Datasheet Criteria
- Watchlist Criteria
- MedDRA Query Criteria
- MedDRA Join
- MedDRA Query
- MedDRA Synonym
- MedDRA Synonym Context
- MedDRA Version
- Object Lifecycle Stage Label
- Object Lifecycle State-Stage
- All Organization objects
- All Person objects
- All Product objects
- Product Alias
- Product Constituent
- Product Family
- Product Registration
- Product Substance
- Product-Event Combination
- PEC Period
- Reason Omitted
- Report
- Reporting Group Product Registration
- Reporting Group Study Registration
- Safety Investigation
- Signal Reporting Period
- Signal Source
- Study
- Study Arm
- Study Arm Product
- Study Indication
- Study Person
- All Study Product objects
- Study Product Group
- Study Product Placeholder
- Study Product Substance
- Study Registration
- Study Site
- Substance
- Substance Alias
- Tab Group
- All Transmission objects
- Unit of Measurement
- All User Task objects
- Validation Result
- Workflow
- Grant Create object permissions on the following objects:
- Product-Event Combination
- PEC Period
- Safety Investigation
- Signal Reporting Period
- Signal Source
- Grant Edit object permissions on the following objects:
- Product-Event Combination
- PEC Period
- Safety Investigation
- Signal Reporting Period
- Signal Source
- Grant Delete object permissions on the PEC Period object.
- Grant View tab permissions for the Signal Management tab collection and the following tabs:
- Home
- Cases
- Analytics
- Reports
- Dashboards
- Literature
- Literature Articles
- Product-Event Combinations
- Safety Investigations
- Safety Investigation Reporting Periods
- Generated PSMFs