You can view all Inbox Items in your Vault on the Inbox tab, which displays all Inbox Items on an object record list page. From here, you can review records created from all case intake methods in a detailed table and access each record to review, process, and promote to Case.
Prerequisites
To support Inbox Items in your Vault, your Admin must enable Inbox Items and assign you permissions to delete data in the Contacts, Products, and Events, sections.
Sort and Filter the Inbox
You can sort and filter the Inbox to quickly review data and identify Inbox Items that require your immediate attention. You can save custom views to return to your view settings.
- Sort: Select any column header to sort the records listed on the page by that option. You can customize the records grid to change the columns displayed on the Inbox page.
- You can sort and filter the records on this page by any standard field on the Inbox Item object to help find and review the applicable records. For example, you can sort by the New Info Date column to view late Inbox Items or filter by Seriousness to view all Inbox Items with adverse events that resulted in death.
- Filter: Select the Edit () icon in the Filters section on the left side of the page to create reusable options. You can select different options within these filters to refine your search even more. You can delete this filter option by selecting the X (
 ) icon in the filter’s header.
) icon in the filter’s header.
Note: Contact your Admin for more information about the standard fields on the Inbox Item object to better understand which fields you can search and filter by on the Inbox tab.
Inbox Item Audit Trail
You can view an audit trail to see the full history of all workflow tasks performed on an Inbox Item. Audit trails include details about the following:
- Inbox Item creation
- Create Case action runs
- User updates any Inbox Item section, including:
- Edit
- Save
- Delete
- Undo (undelete)
Inbox Item States
After you create an Inbox Item, you can change the Inbox Item’s state from the All Actions and Workflow State Change ( ) menus. Some of the default Inbox Item lifecycle states include:
) menus. Some of the default Inbox Item lifecycle states include:
- New: Inbox Item is not yet promoted to a Case.
- Import Error: Inbox Item import has failed. The user who initiated the import can view the error message in the import notification.
- Rejected: Inbox Item rejected during intake. In this state, the Inbox Item is closed to prevent further processing or Case promotion.
- Marked as Follow-Up: Inbox Item is associated with a Case but is not included in that version. You can merge it into the active version or promote it to a new version.
- Marked as Follow-Up (Not Current): Inbox Item generated by the Safety-EDC Connection is associated with a Case that is also associated with a more recent Inbox Item.
- Superseded: Inbox Item is associated with a Case that is also associated with a more recent Inbox Item, which Vault promoted instead of the superseded record.
- Verification: Inbox Item is ready for verification.
- Processing: Inbox Item is in the process of being automatically promoted to an initial or follow-up Case.
- Missing Information: Inbox Item is missing information and cannot be promoted to a Case.
- Duplicate: Inbox Item detected as a duplicate prior to Case promotion. In this state, the Inbox Item is closed to prevent further processing or Case promotion.
- Promoted: Inbox Item successfully promoted to a Case. Vault archives promoted Inbox Items 180 days after the last record update. You cannot modify archived records.
- Promotion Failed: Inbox Item failed automated Case promotion due to not meeting the conditions for being marked as a follow-up Case or created as a follow-up Case.
Data Validation
Vault validates Inbox Item field values during data entry. When you save a record, Vault displays messages so you can correct any missing or invalid data. Additional validations may occur during Case promotion according to your Vault’s configuration. You cannot save Inbox Items or verify Inbox Item sections that do not meet all of the applicable criteria, including values for all required fields and the following:
- Dates: The following rules apply to all date fields:
- Cannot be more than one day in the future. This rule supports data entry in different timezones.
- Must be later than
1800/01/01. - The Date of Death must be later than the First Admin Date, Last Admin Date, Adverse Event Onset and Cessation dates, as well as all dosage administration dates.
- The Last Admin Date must be later than the First Admin Date.
- The Cessation date must be later than the Onset date.
- Minimum precision required for New Info Date is the day.
- Ranks: Only one entry per section can be assigned a Rank of 1 (primary), including Products, Events, and Reporter types of Case Contacts.
- The primary Product must have a value specified in the Product field, as reported by the source.
- Unit Fields: All numeric unit fields must specify both a value and unit, or neither.
- Awareness Details Section: Each Inbox Item must specify New Info Date and Report Type.
- Events Section: Each Event must specify a Type.
- Contacts Section: Each Inbox Item can have only one Patient type of Case Contact. If needed, you can include additional details in the Patient section.
Seriousness and Priority Calculation
Cases should be processed in a timely manner based on their priority. When there is a high volume of Cases, it is crucial to determine Case priority quickly. Vault calculates the Priority and Seriousness of an Inbox Item based on the top-ranked adverse event according to the following order, which you can edit as needed:
| Order | Priority | Seriousness |
|---|---|---|
| 1 | P1 | Results in death |
| 2 | P1 | Life threatening |
| 3 | P2 | Caused / prolonged hospitalisation |
| 4 | P2 | Disabling / incapacitating |
| 5 | P2 | Congenital anomaly / birth defect |
| 6 | P2 | Other medically important condition |
| 7 | P3 | Blank |
Vault considers the following when calculating priority and seriousness:
- Vault assigns P3 priority to Inbox Items without seriousness criteria and one or more associated adverse events.
- Depending on your Admin’s configuration, Required Intervention to Prevent Permanent Impairment/Damage (Devices) may be an available Seriousness value on Cases with Devices and Combination Products. Vault excludes this value when calculating Seriousness and Priority on Inbox Items and Overall Case Seriousness on Cases. When automatically ranking Case Adverse Events, Vault considers this value to be less serious than Other medically important condition, but more serious than a blank Seriousness value.
- When you update the Priority value without changing any other adverse event information, Vault retains the manually updated Priority value. However, if you update, add, or delete the Event, Vault recalculates the Priority upon saving the Inbox Item.
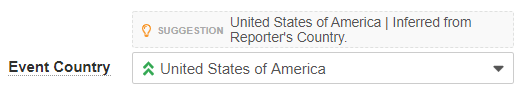
Import Suggestions
For imported Inbox Items, Vault generates suggestions to assist with processing and verification. Vault identifies suggestions with a lightbulb () icon, along with a confidence calculation and rationale, which you can edit as needed. Vault generates the following suggestions:
Product Rank Calculation
Vault calculates and suggests Product rank for imported Inbox Items based on the following:
- Rank 1: Assigned to the first imported Product with a Drug Role of Suspect, Interacting, or Drug Not Administered that matches to a product. If there is no Suspect drug, Vault assigns this rank to the first imported Product with a matching product. Vault assigns this rank to a single record and checks for a product match in the following sequential order:
- Study Product
- Study Product Placeholder
- Company Product
- Rank 2: Assigned to all other Products with matching Study Products, Study Product Placeholders, or Company Products.
- Rank 3: Assigned to all other Products.
Localization
Depending on your Admin’s configuration, Vault may populate the Localization field based on reporter locale:
- If you populate the Site Reporter field of the Inbox Item, Vault populates the Localization based on the reporter’s Country and Reporter Language.
- If your Admin has configured the Enable auto-set Inbox Item Localization by Reporter Country setting, Vault may populate the Localization based on the primary reporter’s locale. Vault references the Reporter type of Case Contact with a Rank of 1:
- If the Country has a related Localization, Vault populates the Localization field based on the Language of the reporter’s Country.
- If the Country has multiple associated Languages, Vault populates the Localization field based on the Reporter Language field of the Case Contact.
- If no match exists, Vault checks for an English record.
- If no English record exists, Vault applies the first Localization record in alphabetical order for that country.
- If configured by your Admin, Vault may populate the Localization based on the the primary reporter’s locale and the report type of the Inbox Item.
When you promote the Inbox Item, Vault processes the Case as domestic or global based on the Localization Type of the matching Localization.
Reporter Locale and Report Type Localization
When configured by your Admin, Vault populates the Localization field on imported Inbox Items based on the primrary reporter’s locale and the report type of the Inbox Item. Vault references the Reporter type of Case Contact with a Rank of 1 and the Report Type of the Inbox Item. When the Report Type is Study, Vault also references the Study Type of the associated Study. Vault matches Localization based on its Case Type values as follows:
- Blank: Matches regardless of the Report Type of the Inbox Item.
- Investigational: Matches if the Report Type of the Inbox Item maps to E2B code
2(Report in Clinical Trial) and the Study Type of the Study maps to E2B code1(Clinical Trial), or Study Type is blank. For example, the Study Type is Clinical Trial. - Marketing: Matches if either of the following conditions are true:
- The Report Type of the Inbox Item maps to any E2B code except
2(Report in Clinical Trial). For example, when the Report Type is any value other than Study. - The Report Type of the Inbox Item maps to E2B code
2(Report in Clinical Trial) and the Study Type of the Study maps to any E2B code except1. For example, when the Study Type is any value other than Clinical Trial.
- The Report Type of the Inbox Item maps to any E2B code except
Vault then populates the Localization field based on the matching Localization record:
- If one match for the primary reporter’s Country and Report Type exists, Vault populates the field with the corresponding Localization value.
- If multiple matches for the primary reporter’s Country and Report Type exist, Vault references the Reporter Language field of the Case Contact.
- If Reporter Language is blank, Vault checks for an English record.
- If there are still multiple matches, Vault applies the first Localization record for that Country or Report Type.
- If zero matches for the primary reporter’s Country and Report Type exist, Vault does not populate the field.