Learn how Vault generates the October 2015 version of the FDA MedWatch 3500A form from a Case. For details about the available FDA MedWatch 3500A formats supported by Safety and how Vault handles certain data during form generation, see FDA Report Formats.
Header
| FDA MedWatch 3500A Field | Populated Value |
|---|---|
| Mfr. Report # | The value from the UID field on the Case. |
A. Patient Information
| FDA MedWatch 3500A Field | Populated Value |
|---|---|
| 1. Patient Identifier | The Patient Initials / ID on the Case. If blank, Vault populates the Investigation MRN value. |
| 2. Age | The value from one of the following Case fields, ordered by priority:
|
| 2. Date of Birth | The Date of Birth on the Case. |
| 3. Sex | The Sex on the Case. |
| 4. Weight | The value from one of the following Case fields, ordered by priority:
|
| 5.a. Ethnicity | The Ethnicity on the Case. |
| 5.b. Race | The Race on the Case. |
B. Adverse Event or Product Problem
| FDA MedWatch 3500A Field | Populated Value |
|---|---|
| 1. Type of Report | Based on the report type, Vault selects:
|
| 2. Outcome Attributed to Adverse Event | Vault selects the appropriate checkbox based on the Seriousness of the Case. If the adverse event resulted in death, Vault populates the Date of Death on the Case. |
| 3. Date of Event | The earliest Onset Date for any Case Adverse Event associated with the Case. |
| 4. Date of this Report | The date when the report was generated, according to the Vault's timezone. |
| 5. Describe Event or Problem | Any text in the Narrative on the Case. After the narrative text, Vault populates any text in the Company Comments field. If the narrative text overflows onto additional pages, Company Comments also appear on the overflow pages. |
| 6. Relevant Tests/Laboratory Data, Including Dates | On each row, Vault populates a Case Test Result. Records appear alphabetically and from oldest to latest. The information exported includes:
In addition, if the adverse event resulted in death, Vault populates the following Case Cause of Death information:
|
| 7. Other Relevant History, Including Preexisting Medical Conditions | For each Case Medical History record, Vault populates:
After listing each medical history line item, Vault populates any Medical History Text on the Case. |
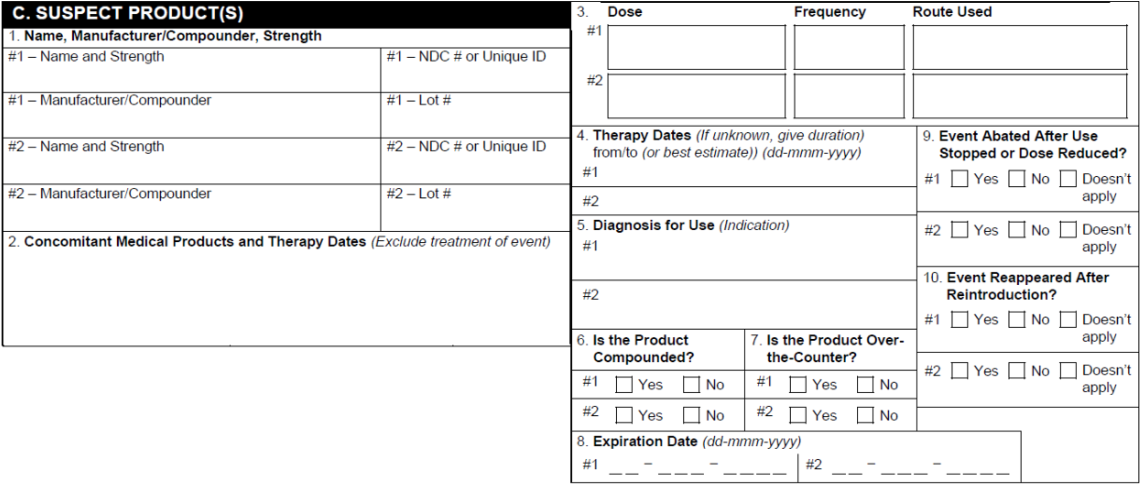
C. Suspect Products
| FDA MedWatch 3500A Field | Populated Value | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Name and Strength, Manufacturer/Compounder | For Case Products assigned a Drug Role of Suspect, Interacting, or Drug Not Administered, Vault populates:
|
||||||||||||||||||||||||||||||||
| 2. Concomitant Medical Products and Therapy Dates | For each Case Product, Vault populates:
If the therapy dates are unknown, Vault populates the Duration on the Case Product Dosage. Vault populates
Note: For device-only Cases, this information is located in Section D.11 below. |
||||||||||||||||||||||||||||||||
| 3. Dose, Frequency, Route Used | For Case Products assigned a Drug Role of Suspect, Interacting, or Drug Not Administered, Vault populates:
|
||||||||||||||||||||||||||||||||
| 4. Therapy Dates | Vault populates Case Product information using the following priority:
|
||||||||||||||||||||||||||||||||
| 5. Diagnosis for Use | The Name (MedDRA) or Indication (Reported) on the Case Product Indication. | ||||||||||||||||||||||||||||||||
| 6. Is the Product Compounded? | If any value is selected in the Compounding Type field on the Case Product Registration, then the Compounded checkbox is selected. | ||||||||||||||||||||||||||||||||
| 7. Is the Product Over-the-Counter? | For each Case Product section, if any part of the Product has an FDA registration with the Registered As field set to a Transmission Product Type of OTC Drug, the Yes checkbox is selected. Otherwise, the field is left blank. | ||||||||||||||||||||||||||||||||
| 8. Expiration Date | The Expiration Date on the Case Product. | ||||||||||||||||||||||||||||||||
| 9. Event Abated After Use Stopped or Dose Reduced? | Vault calculates whether a dechallenge test occurred using the following fields:
Based on the field values, Vault populates the form as follows:
|
||||||||||||||||||||||||||||||||
| 10. Event Reappeared After Reintroduction? | Vault uses the Reaction Recurrence field on the primary Case Assessment to determine whether a rechallenge test occurred and populate the appropriate checkbox.
|
||||||||||||||||||||||||||||||||
D. Suspect Medical Device
| FDA MedWatch 3500A Field | Populated Value |
|---|---|
| 1. Brand Name | The Product (Reported) on the Device-type Case Product. Note: Your Admin can configure your Vault to export the Product (Coded) field by default. |
| 2. Common Device Name | The Generic Name on the associated Product. |
| 2b. Procode | The Product Code on the associated Product Registration. |
| 3. Manufacturer Name, City and State | If the Manufacturer field on the associated Product Family is an Organization, Vault populates the associated name, city, and state. Otherwise, Vault populates the Organization of the associated Product Family. |
| 4. Model # | The Model Number on the Device-type Case Product. |
| 4. Lot # | The Lot Number on the Device-type Case Product. |
| 4. Catalog # | The Catalog Number on the Device-type Case Product. |
| 4. Expiration Date | The Expiration Date on the Device-type Case Product. |
| 4. Serial # | The Serial Number on the Device-type Case Product. |
| 4. Unique Identifier (UDI) # | The Unique Identifier on the Device-type Case Product. |
| 5. Operator of Device | The Operator of Device on the Device-type Case Product. |
| 6. If Implanted, Give Date | The Date Implanted on the Device-type Case Product. |
| 7. If Explanted, Give Date | The Date Explanted on the Device-type Case Product. |
| 8. Is this a single-use device that was reprocessed and reused on a patient? | Vault selects the appropriate checkbox based on the Reprocessed/Reused field on the Device-type Case Product. |
| 9. If Yes to Item 8, Enter Name and Address of Reprocessor | The Reprocessor value on the Device-type Case Product. |
| 10. Device Available for Evaluation? | Vault selects the appropriate checkbox based on the Device Available field on the Device-type Case Product. If there is a Returned Date, Vault populates the date and selects the Returned to Manufacturer checkbox. |
| 11. Concomitant Medical Products and Therapy Dates |
For each Case Product, Vault populates values from the following fields:
If the therapy dates are unknown, Vault populates the Duration on the Case Product Dosage. Vault populates
|
E. Initial Reporter
| FDA MedWatch 3500A Field | Populated Value |
|---|---|
| 1. Name and Address | The name and address on the primary Reporter-type Case Contact. |
| 2. Health Professional? | Vault selects the appropriate checkbox based on the Qualification on the primary Reporter-type Case Contact. |
| 3. Occupation | The Qualification on the primary Reporter-type Case Contact. |
| 4. Initial Reporter Also Sent Report to FDA | Vault selects the appropriate checkbox based on the Sent to FDA? field on the primary Reporter-type Case Contact. |
F. For Use by User Facility/Importer (Devices Only)
Note: Vault does not populate section F, but the section is available on the form for manual data entry.
G. All Manufacturers
| FDA MedWatch 3500A Field | Populated Value |
|---|---|
| 1. Contact Office (and Manufacturing Site for Devices) or Compounding Outsourcing Facility | The contact information for the Sender User on the associated Transmission (Submission or Distribution). When generating a form preview from a Case, Vault populates contact details for the Organization on the Case. |
| 2. Phone Number | When generating a form for submission or distribution, Vault populates the phone number for the user selected in the Sender User field on the associated Transmission (Submission or Distribution) record. When generating a form preview from the Case, Vault populates the phone number for the organization selected in the Manufacturer on the Product record associated with the Case Product. |
| 3. Report Source | Vault selects the appropriate checkboxes as follows:
|
| 4. Date Received by Manufacturer | Vault uses the following logic to populate this field: Initial Case: Vault maps the date from one of the following Case fields, ordered by priority:
A Case is considered initial when it meets any of the following conditions:
Follow-Up: Vault maps the date from the New Info Date field on the Case, if populated. A Case is considered follow-up when it meets any of the following conditions:
|
| 5. NDA # | If the Registration Type on the associated Product Registration is NDA, Vault maps the Registration Number. |
| 5. ANDA # | If the Registration Type on the associated Product Registration is ANDA, Vault maps the Registration Number. |
| 5. IND # | For study Cases with a Study Type of Clinical Trial or blank, if there is a US Study Registration with a Registration Type of IND, Vault populates the IND number from the Study Registration Number field from that Study Registration. In all other scenarios, if the Registration Type on the Case Product Registration is IND, Vault populates the Registration Number. |
| 5. BLA # | If the Registration Type on the associated Product Registration is BLA, Vault maps the Registration Number. |
| 5. PMA/510(k) # | If the Registration Type on the associated Product Registration is either PMA or 510k, Vault maps the Registration Number. |
| 5. Combination Product | Vault selects this checkbox when any Case Product is part of a Combination Product.
Note: For study Cases with Products that are part of a Combination Product, if Study Content Protection is enabled or a blinded regulatory report preview is generated, Vault does not select this checkbox. |
| 5. Pre-1938 | If the International Birthdate on the associated Product is earlier than 1938, Vault selects this checkbox. |
| 5. OTC | Across all Case Products in sections C and D, if any part of a Product has an FDA registration with the Registered As field set to a Transmission Product Type of OTC Drug or OTC Device, Vault selects the Yes checkbox. |
| 6. If IND/PreANDA, Give Protocol # | The Sponsor Study Number on the Case. |
| 7. Type of Report | Vault selects the appropriate checkbox based on the value in the FDA Report Type field, which is set based on the Due in Days reporting rule parameter. Otherwise, Vault selects checkboxes based on the following logic:
When no report type applies based on the above criteria, Vault selects the Periodic checkbox. Vault populates the Follow-up # value from the Follow-up Number field on the Submission record. |
| 8. Adverse Event Term(s) | The Event (MedDRA) or the Event (Reported) - English on the primary Case Adverse Event. |
| 9. Manufacturer Report Number | The UID on the Case. |
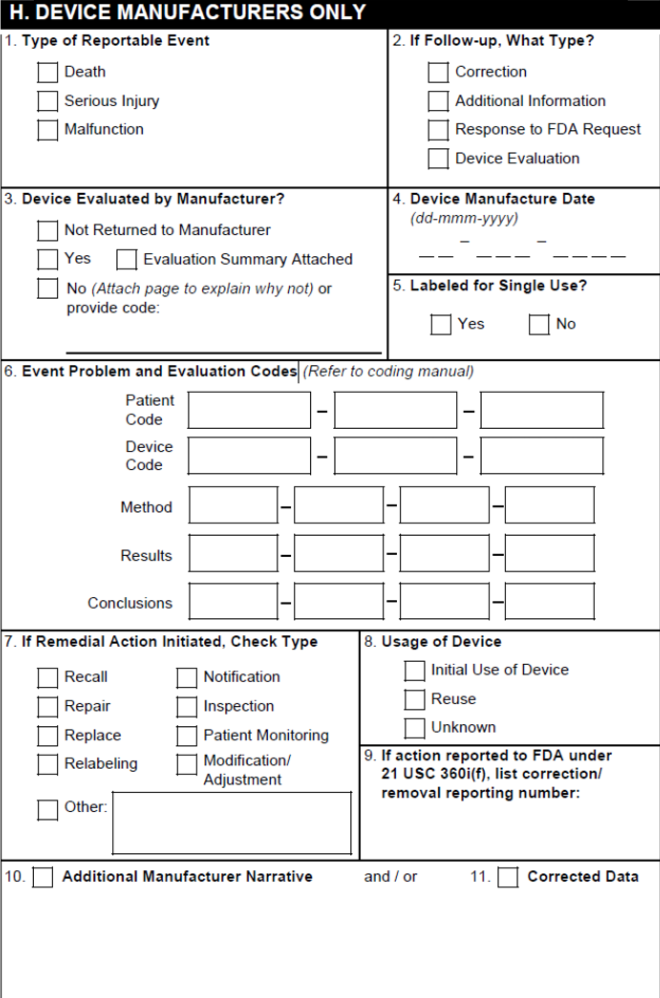
H. Device Manufacturers Only
Note: Section H is populated only for Cases concerning a Combination Product with a Device-type Product Constituent.
| FDA MedWatch 3500A Field | Populated Value |
|---|---|
| 1. Type of Reportable Event | Vault selects the Death checkbox if the Seriousness of any Case Adverse Event contains Results in death. Vault selects the Serious Injury checkbox based on the value in the Device Report Type field on the Case. Vault selects the Malfunction checkbox based on the Malfunction field on the primary Device-type Case Product. If that field is blank, Vault selects a checkbox based on the Device Report Type on the Case. |
| 2. If Follow-up, What Type? | Vault selects the appropriate checkboxes based on the Device Follow-Up Type on the Case. |
| 3. Device Evaluated by Manufacturer | Vault selects the appropriate checkbox based on the Device Evaluated value on the Case Product. If Yes is selected, the Evaluation Summary Attached checkbox is also selected. |
| 4. Device Manufacture Date | The Manufacture Date on the Case Product. |
| 5. Labeled for Single Use? | For Device-type Case Products, the Single Use value. |
| 6. Event Problem and Evaluation Codes | Codes in this section are populated as follows:
To use IMDRF codes, your Admin must enable the IMDRF Dictionary for Maintenance, Case Processing, and Submissions. |
| 7. Remedial Action Initiated, Check Type | Vault selects the appropriate boxes using the following priority order:
|
| 8. Usage of Device | Vault selects the appropriate box based on the Device Usage Type on the Device-type Case Product. |
| 9. If action reported to FDA under 21 USC 360i(f), list correction/removal reporting number | The Correction/Removal Reporting Number on the Device-type Case Product. |
| 10. Additional Manufacturer Narrative | The Additional Manufacturer Narrative on the Device-type Case Product. Text that exceeds the field character limit appears in the overflow pages under the heading H.10 Additional Manufacturer Narrative. |
| 11. Corrected Data | This field is not automatically populated. |
Note: If your organization is not using Case Product-level device coding, Vault exports FDA codes for one (1) device. This is either the highest-ranked or earliest-entered device on the Case. The code entered on the Case is populated for each of the following:
- Device Problem
- Evaluation Method
- Evaluation Result
- Evaluation Conclusion
Overflow Pages
When generating the FDA MedWatch 3500A form, Vault exports any text that exceeds field character limits to overflow pages. In the applicable field, Vault truncates the text to 15 characters and appends (Continued) to indicate the information is on the overflow pages. Any text on the overflow pages is identified with the relevant section and field names.